- Laboratory
- Laboratory medicine
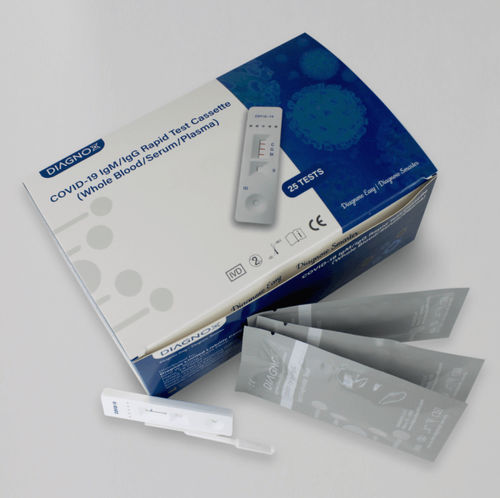
- COVID-19 test kit
- Diagnox Health
COVID-19 test kit IgGIgMSARS-COV-2
Add to favorites
Compare this product
Characteristics
- Applications
- COVID-19
- Tested parameter
- IgG, IgM
- Micro-organism
- SARS-COV-2
- Sample type
- serum, plasma, whole blood
- Analysis mode
- lateral flow
- Format
- cassette
Description
The COVID-19 Antibodies test from Diagnox is an FDA-EUA, C-FDA, CE-IVD, and A-TGA approved test for IgG and IgM antibodies. It is a rapid test device that utilizes lateral flow technology for the qualitative, differential detection of both anti-SARS-CoV-2 IgM (early marker) and IgG (late marker) antibodies. The test works with whole blood, serum, or plasma.
Features
Facilitates patient treatment decisions quickly
Detection window (IgM): 3-5 days for Symptomatic. 7 days for Asymptomatic
No clinical equipment or facilities needed
Dual-band results for simple interpretation
Multivariable analysis of immunoglobin IgG & IgM
Specifications
Format: Cassette
Time to results: 10 minutes
Specimen: Whole Blood/Serum/Plasma
Volume: 10 uL
Positive Percent Agreement: IgG 96.7% | IgM 86.7% | Overall 96.7%
Negative Percent Agreement: IgG 98.0% | IgM 99.0% | Overall 97.0%
Shelf life: 24 Months
Storage: Room temperature or refrigerated (2-30⁰C / 36-86⁰F)
Catalogs
Related Searches
- Assay kit
- Blood assay kit
- Serum assay kit
- Plasma assay kit
- Rapid lateral flow test
- Respiratory infection test kit
- Whole blood detection kit
- Cassette assay kit
- Lateral flow test kit
- COVID-19 assay kit
- Urine rapid diagnostic test
- IgG test kit
- Test strip
- Urine assay kit
- Rapid drug abuse test
- Self-test assay kit
- Urine test strip
- Abused drug test kit
- Protein test strip
- PH test strip
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.



