- Laboratory
- Laboratory medicine
- COVID-19 rapid test
- Hangzhou GENESIS Biodetection and Biocontrol CO., LTD

- Products
- Catalogs
- News & Trends
- Exhibitions
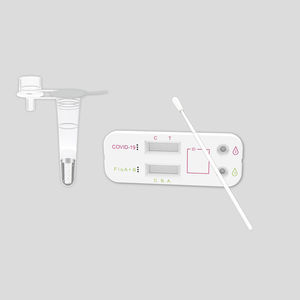
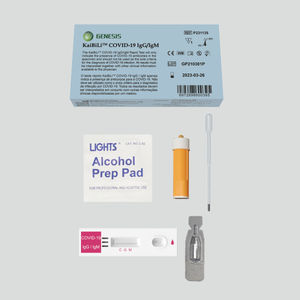
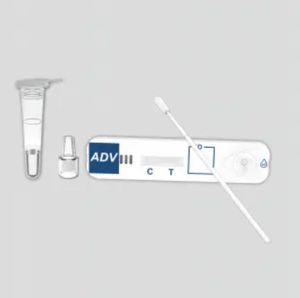
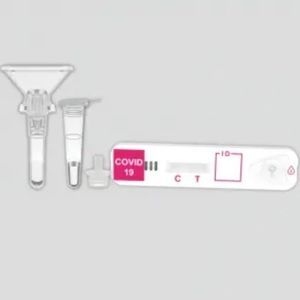
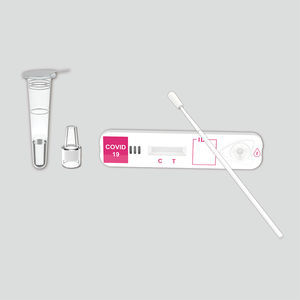
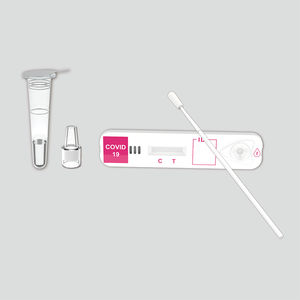
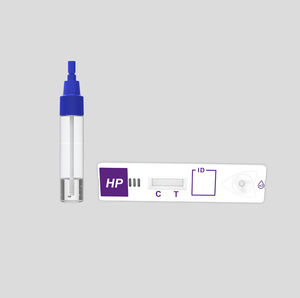
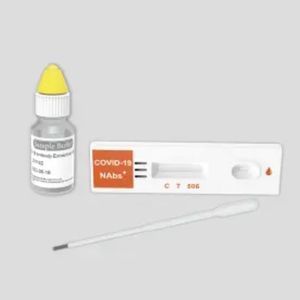
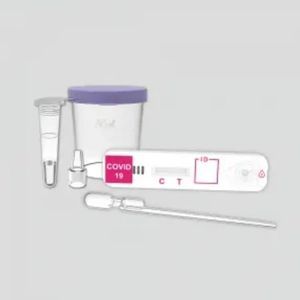
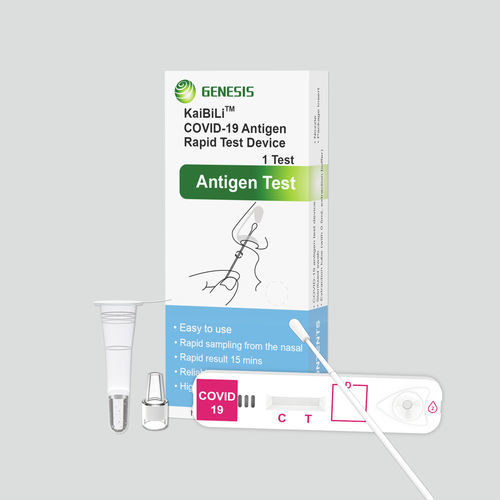
COVID-19 rapid test KaiBiLi™for antigensSARS-COV-2coronavirus
Add to favorites
Compare this product
Characteristics
- Applications
- COVID-19
- Tested parameter
- for antigens
- Micro-organism
- SARS-COV-2, coronavirus
- Sample type
- nasal
- Analysis mode
- immunoassay
- Result display time
15 min, 30 min
Description
The KaiBiLiTM COVID-19 Antigen is an in vitro diagnostic test based on the principle of immuno- chromatography for the qualitative detection of 2019 Novel Coronavirus nucleocapsid protein antigens in nasal swabs.
The KaiBiLiTM COVID-19 Antigen is an in vitro diagnostic test based on the principle of immuno- chromatography for the qualitative detection of 2019 Novel Coronavirus nucleocapsid protein antigens in nasal swabs. The detection is based on the antibodies which were developed specifically recognizing and reacting with the nucleoprotein of 2019 Novel Coronavirus. It is intended to aid in the rapid diagnosis of SARS-CoV-2 infection. Positive results indicate the presence of viral antigens, but clinical correlation with patient history and other diagnostic information is necessary to determine infection status.
Donors with a negative result should be treated as presumptive. Negative results do not rule out SARS-CoV-2 infection and should not be used as the sole basis for treatment or patient management decisions, including infection control decisions.
COVID-19 is an acute respiratory infectious disease. People are generally susceptible. Currently, the patients infected by the novel coronavirus are the main source of infection; asymptomatic infected people can also be an infectious source. Based on the current epidemiological investigation, the incubation period is 1 to 14 days, mostly 3 to 7 days. The main symptoms including manifestations include fever, fatigue and dry cough, nasal congestion, runny nose, sore throat, myalgia and diarrhea are found in a few cases.
The qualitative detection of 2019 Novel Coronavirus nucleocapsid protein antigens in nasal swabs.
Other Hangzhou GENESIS Biodetection and Biocontrol CO., LTD products
IMMUNOASSAYS
Related Searches
- Assay kit
- Solution reagent kit
- Immunoassay assay kit
- Infectious disease detection kit
- Blood rapid diagnostic test
- Diagnostic reagent kit
- Rapid lateral flow test
- Laboratory reagent kit
- Immunoassay rapid diagnostic test
- Molecular test kit
- Rapid virus test
- Respiratory infection test kit
- Serum rapid diagnostic test
- Plasma rapid diagnostic test
- Reagent medium reagent kit
- Infectious disease rapid diagnostic test
- Whole blood rapid diagnostic test
- Buffer solution reagent kit
- Lateral flow test kit
- COVID-19 detection kit
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.