- Laboratory
- Laboratory medicine
- Infectious disease test kit
- Hebei Guogao Biotechnology

- Products
- Catalogs
- News & Trends
- Exhibitions
SARS-COV-2 assay kit for antigensIgGcoronavirus
Add to favorites
Compare this product
Characteristics
- Tested parameter
- for antigens, IgG
- Micro-organism
- SARS-COV-2, coronavirus
- Sample type
- clinical, laboratory, nasal, throat
- Analysis mode
- immunochromatographic
- Result display time
15 min
- Specificity
98.3 %
- Sensitivity
92.7 %
Description
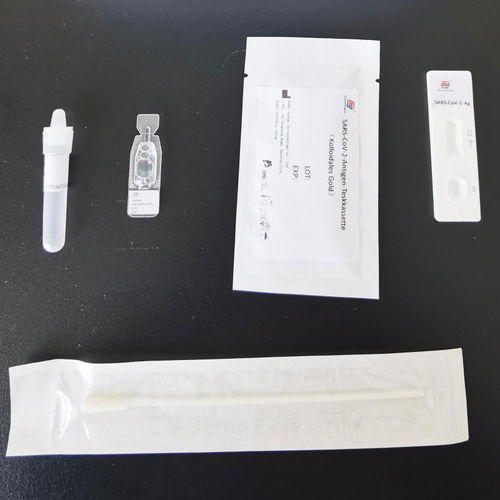
This product is used to qualitatively detect the novel coronavirus (SARS-CoV-2) antigen in human nasal throat swabs or oral throat swabs.
It is only suitable for professional in vitro diagnosis, not for personal use.
This product is only used in clinical laboratories or immediate testing by medical staff. It cannot be used for home testing. It cannot be used as the basis for diagnosis and exclusion of pneumonia caused by new coronavirus
infections. It is not suitable for screening by the general population.
A positive test result requires further confirmation, and a negative test result cannot rule out the possibility of infection.
The kit and test results are for clinical reference only. It is recommended that the patient's clinical manifestations and other laboratory examinations be combined for a comprehensive analysis of the condition.
The kit cannot distinguish between SARS-CoV and SARS-CoV-2.
[Test Principle]
This product adopts colloidal gold immunochromatography technology, spraying colloidal gold labeled SARS-CoV-2 monoclonal antibody 1 on the gold pad The SARS-CoV-2 monoclonal antibody 2 is coated on nitrocellulose membrane as the test line (T line) and goat anti-mouse IgG antibody is coated as the quality control line (C line). When an appropriate amount of the sample to be tested is added to the sample hole of the test card, the sample will move forward along the test card under capillary action.
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.
