Respiratory disease test kit RIDA®GENE SARS-COV-2adenovirusenterovirus
Add to favorites
Compare this product
Characteristics
- Applications
- for respiratory diseases
- Micro-organism
- SARS-COV-2, adenovirus, enterovirus
- Sample type
- clinical, laboratory, nasal, throat
- Analysis mode
- for real-time PCR
Description
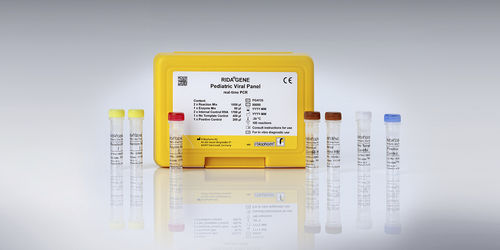
For in vitro diagnostic use. The RIDA®GENE Pediatric Viral Panel test, performed on the LightCycler® 480 II real-time PCR instrument, is a multiplex real-time RT-PCR for the direct qualitative detection and differentiation of rhinovirus/ enterovirus/ parechovirus RNA, bocavirus DNA, and adenovirus DNA in untreated human nasal/throat swabs from people with signs and symptoms of a respiratory infection.
The RIDA®GENE Pediatric Viral Panel test is intended to support the differential diagnosis of rhinovirus/enterovirus/parechovirus, bocavirus, and adenovirus infections in patients with symptoms of a respiratory infection in connection with other clinical and laboratory findings.
Negative results do not rule out infection with rhinovirus/enterovirus/parechovirus, bocavirus, or adenovirus and should not be used as the sole basis for diagnosis.
The product is intended for professional use.
General information:
Viral respiratory tract infections are one of the primary causes of hospitalizations and clinic visits in children and older people. In addition to the known respiratory viruses like influenza and SARS-CoV-2, adenoviruses, bocaviruses, enteroviruses, parechoviruses, and rhinoviruses can cause respiratory tract infections.
Human adenoviruses (HAdVs) belong to the family Adenoviridae and the genus Mastadenovirus. They are icosahedron-shaped, nonenveloped, double-stranded DNA viruses with a diameter of 65 to 110 nm. They are divided into seven species, (A to G), which comprise about 67 serotypes.
Catalogs
No catalogs are available for this product.
See all of R-Biopharm AG‘s catalogsRelated Searches
- Assay kit
- Solution reagent kit
- Blood assay kit
- Serum assay kit
- Immunoassay assay kit
- Plasma assay kit
- Infectious disease detection kit
- Rapid lateral flow test
- Diagnostic reagent kit
- Immunoassay rapid diagnostic test
- Molecular test kit
- Respiratory infection test kit
- Infectious disease rapid diagnostic test
- Clinical assay kit
- Cassette assay kit
- Lateral flow test kit
- ELISA assay kit
- COVID-19 detection kit
- Real-time PCR test kit
- Quality control reagent kit
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.