- Laboratory
- Laboratory medicine
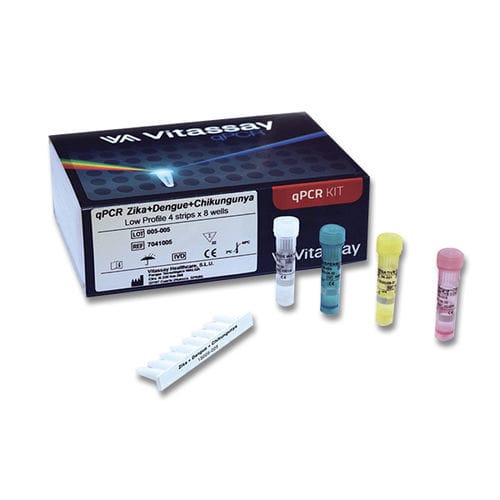
- Chikungunya test kit
- VITASSAY HEALTHCARE S.L.
Chikungunya test kit 7041001virusclinicalfor RT-PCR
Add to favorites
Compare this product
Characteristics
- Applications
- chikungunya
- Micro-organism
- virus
- Sample type
- clinical
- Analysis mode
- for RT-PCR
Description
Vitassay qPCR Chikungunya allows the qualitative detection of Chikungunya virus by
real-time RT-PCR in clinical samples. The product is intended for use in the diagnosis of
Chikungunya virus infections alongside clinical data of the patient and other laboratory
tests outcomes.
Transport and storage
• The reagents and the test can be shipped and stored at 2-40ºC until expiration
date stated in the label.
• The resuspended positive control should be stored at -20ºC. In order to avoid
repeated freeze/thaw cycles, it is recommended to distribute the content in
different aliquots.
• Keep all reagents in the darkness.
Additional equipment and material required
• Real-time PCR instrument (thermocycler) (Attached I)
• RNA extraction kit
• Centrifuge for 1.5 mL tubes
• Vortexer
• Micropipettes (1-20 µL, 20-200 µL)
• Filter tips
• Powder-free disposal gloves
Summary
Chikungunya virus (CHIKV) is a mosquito-borne virus (arbovirus) first described during
an outbreak in southern Tanzania in 1952. It’s transmitted to humans mainly by infected
mosquitos of the Aedes genus, specifically Ae. aegypti and Ae. albopictus. Although
perinatal vertical transmission has also been reported.
Since 1952, CHIKV has caused numerous well-documented outbreaks and epidemics in
Africa, Asia, Europe, the South Pacific and Americas, involving millions of people, and
often separated by periods of more than 10 years.
Catalogs
Vitassay_Catalogue
20 Pages
Related Searches
- Assay kit
- Solution reagent kit
- Blood assay kit
- Molecular biology reagent kit
- Plasma assay kit
- Infectious disease detection kit
- Blood rapid diagnostic test
- Diagnostic reagent kit
- Rapid lateral flow test
- Enzyme reagent kit
- Immunoassay rapid diagnostic test
- Molecular test kit
- Cassette rapid diagnostic test
- Virus rapid diagnostic test
- Respiratory infection test kit
- Optical assay kit
- Clinical assay kit
- Infectious disease rapid diagnostic test
- Buffer solution reagent kit
- Fluorescence assay kit
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.